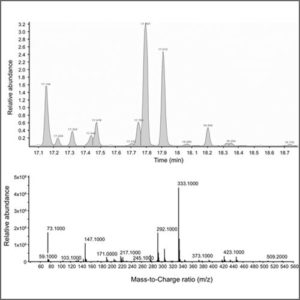
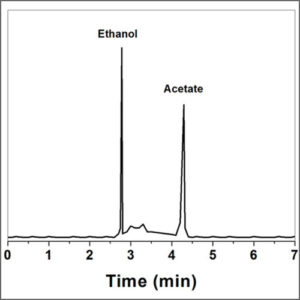
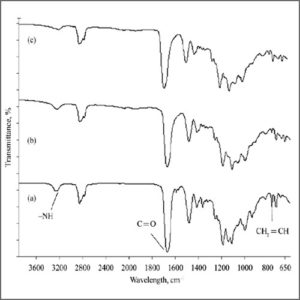
Freeze Dryer
Freeze dryer is widely used in dehydration process in order to preserve a perishable material or make the material convenient for transport.
- Description
| Testing Method | Freeze Dryer |
| Description |
Freeze dryer is widely used in dehydration process in order to preserve a perishable material or make the material convenient for transport. Freeze-drying works by freezing the material and then reducing the surrounding pressure to allow the frozen water in the material to sublimate directly from the solid phase to the gas phase.
There are four stages in the complete drying process: pre-treatment, freezing, primary drying, and secondary drying. Pre-treatment includes any method of treating the product prior to freezing. Freezing is often done by placing the material in a freeze-drying flask and rotating the flask in a bath, called a shell freezer, which is cooled by mechanical refrigeration, dry ice and methanol, or liquid nitrogen. During the primary drying phase, the pressure is lowered (to the range of a few milli-bars), and enough heat is supplied to the material for the ice to sublime. The secondary drying phase aims to remove unfrozen water molecules, since the ice was removed in the primary drying phase.
If a freeze-dried substance is sealed to prevent the reabsorption of moisture, the substance may be stored at room temperature without refrigeration, and be protected against spoilage for many years. Preservation is possible because the greatly reduced water content inhibits the action of microorganisms and enzymes that would normally spoil or degrade the substance. |
| More Information | Wikipedia: Freeze Drying |